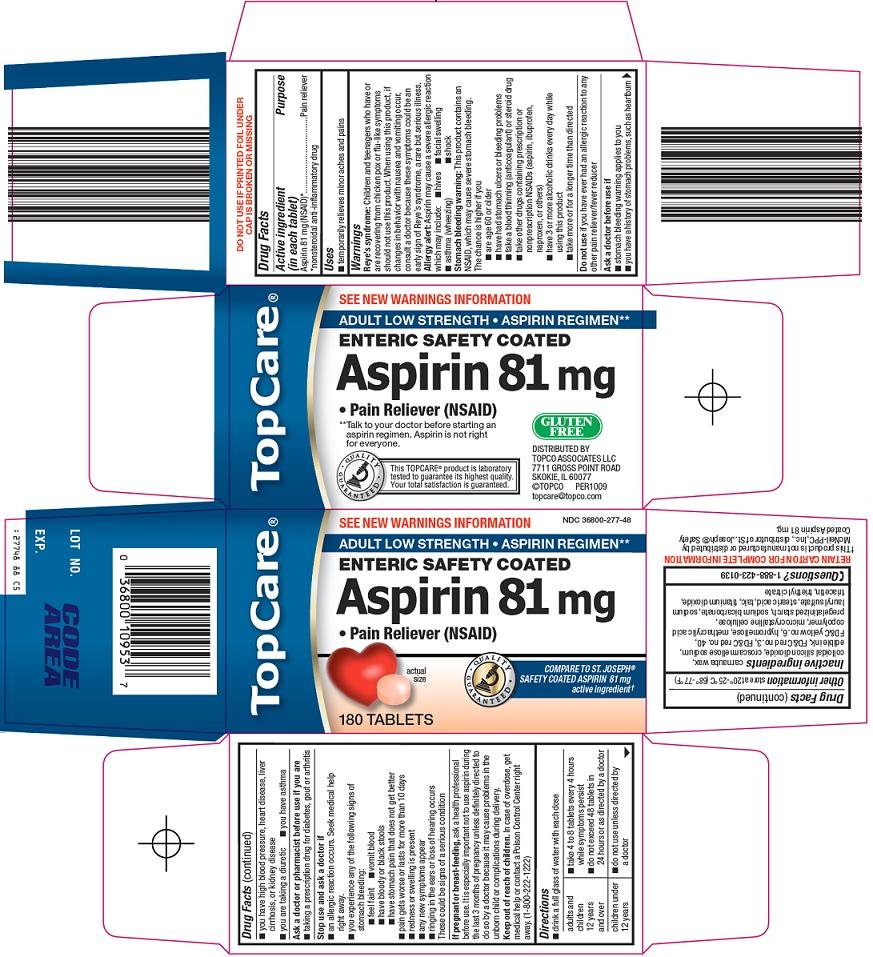
TOPCARE ASPIRIN
-
aspirin tablet, film coated
Topco Associates LLC
Aspirin 81 mg (NSAID)*
*nonsteroidal anti-inflammatory drug
Pain reliever
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding.
The chance is higher if you
if you have ever had an allergic reaction to any other pain reliever/fever reducer
These could be signs of a serious condition
ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
| adults and children 12 years and over |
|
| children under 12 years |
|
store at 20º-25ºC (68º-77ºF)
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, edible ink, FD&C red no. 3, FD&C red no. 40, FD&C yellow no. 6, hypromellose, methacrylic acid copolymer, microcrystalline cellulose, pregelatinized starch, sodium bicarbonate, sodium lauryl sulfate, stearic acid, talc, titanium dioxide, triacetin, triethyl citrate
1-888-423-0139
See New Warnings Information
Adult Low Strength
Aspirin Regimen**
Enteric Safety Coated
Aspirin 81 mg
Pain Reliever (NSAID)
Actual Size
Compare to St.Joseph® Safety Coated Aspirin 81 mg active ingredient
Aspirin 81 mg Carton
|
TOPCARE ASPIRIN
aspirin tablet, film coated | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part343 | 05/03/2023 | |
| Labeler - Topco Associates LLC (006935977) |