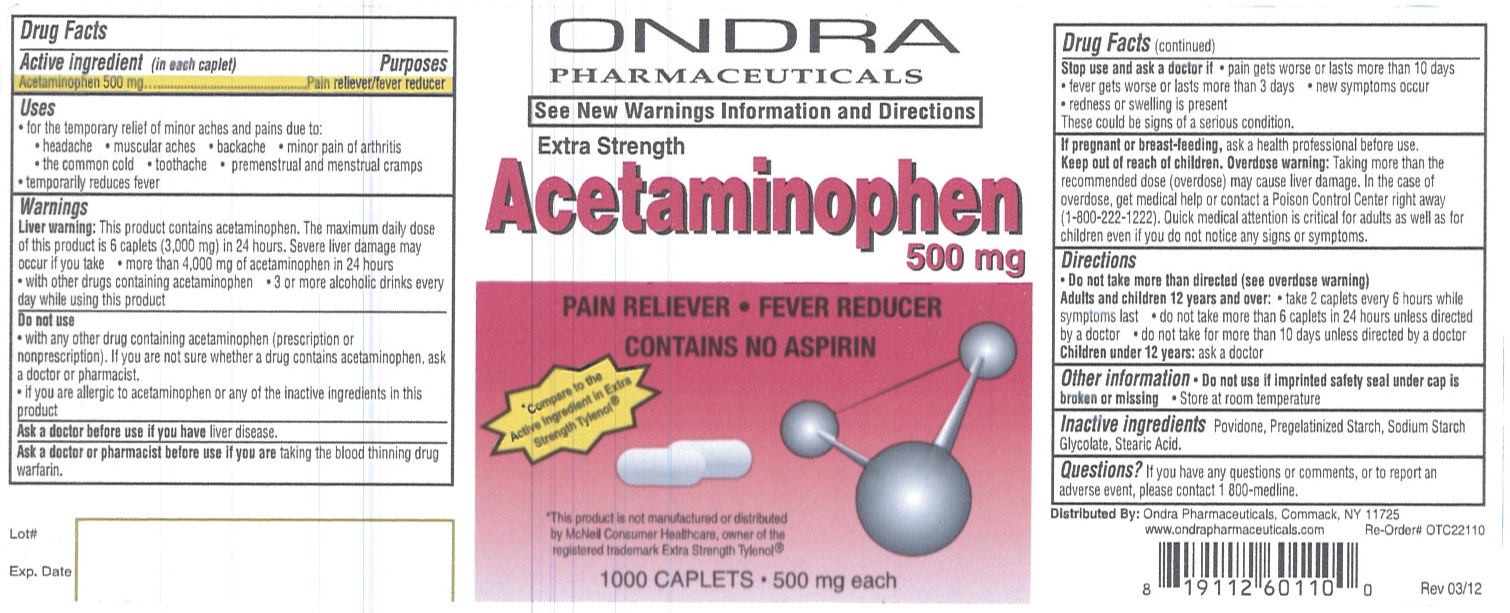
ACETAMINOPHEN
-
acetaminophen tablet
Gemini Pharmaceuticals, Inc. dba ONDRA Pharmaceuticals
Acetaminophen 500 mg
Pain reliever/fever reducer
Liver warning: This product contains acetaminophen. The maximum daily dose of this product is 6 caplets (3,000 mg) in 24 hours. Severe liver damage may occur if you take
liver disease.
taking the blood thinning drug warfarin.
If pregnant or breast-feeding, ask a health professional before use.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In the case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Adults and children 12 years and over:
Children under 12 years: ask a doctor
Povidone, Pregelatinized Starch, Sodium Starch Glycolate, Stearic Acid.
If you have any questions or comments, or to report an adverse event, please contact 1 800-medline.
|
ACETAMINOPHEN
acetaminophen tablet | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part343 | 03/30/2012 | |
| Labeler - Gemini Pharmaceuticals, Inc. dba ONDRA Pharmaceuticals (055942270) |
| Registrant - Gemini Pharmaceuticals, Inc. dba ONDRA Pharmaceuticals (055942270) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Gemini Pharmaceuticals, Inc. dba ONDRA Pharmaceuticals | 055942270 | manufacture | |