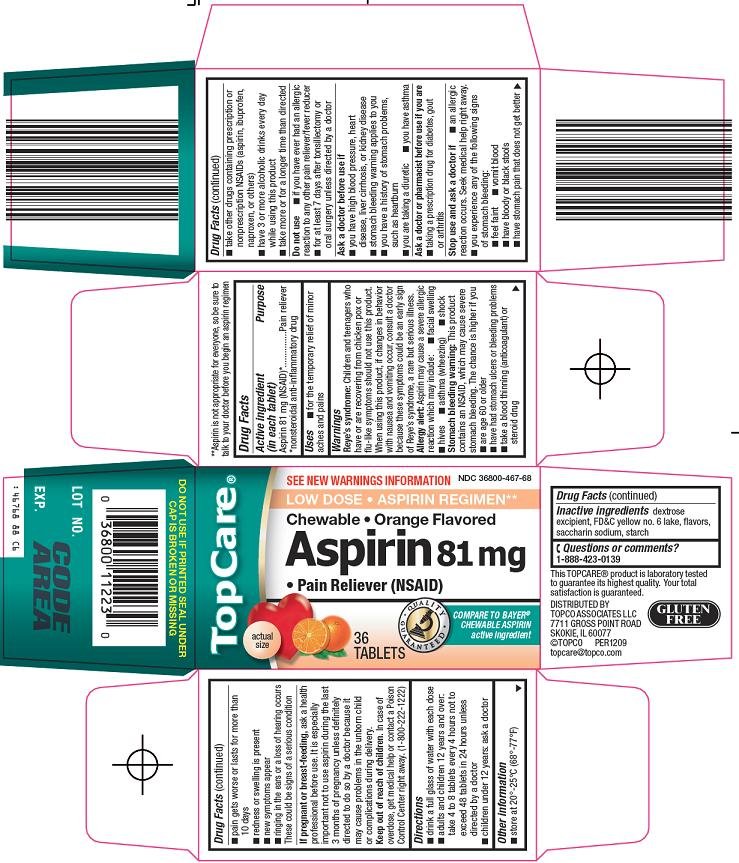
TOPCARE ASPIRIN
-
aspirin tablet, chewable
Topco Associates LLC
Topco Aspirin 81 mg Drug FactsActive ingredient (in each tablet)
Aspirin 81 mg (NSAID)*
*nonsteroidal anti-inflammatory drug
Purpose
Pain reliever
Uses
- for the temporary relief of minor aches and pains
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Do not use
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- for at least 7 days after tonsillectomy or oral surgery unless directed by a doctor
Ask a doctor before use if
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you are taking a diuretic
- you have asthma
Ask a doctor or pharmacist before use if you are
- taking a prescription drug for diabetes, gout or arthritis
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts for more than 10 days
- redness or swelling is present
- new symptoms appear
- ringing in the ears or a loss of hearing occurs
These could be signs of a serious condition
If pregnant or breast-feeding,
ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Directions
- drink a full glass of water with each dose
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- children under 12 years: ask a doctor
Other information
- store at 20°-25°C (68°-77°F)
Inactive ingredients
dextrose excipient, FD&C yellow no. 6 lake, flavors, saccharin sodium, starch
Questions or comments?
1-888-423-0139
Principal Display Panel
See New Warnings Information
Low Dose
Aspirin Regimen**
Chewable
Orange Flavored
Aspirin 81 mg
Pain Reliever (NSAID)
Actual Size
Compare to Bayer® Chewable Aspirin active ingredient
Aspirin 81 mg Carton
TOPCARE ASPIRIN
aspirin
tablet, chewable
|
|
|
|
|
|
Revised: 04/2010Topco Associates LLC
