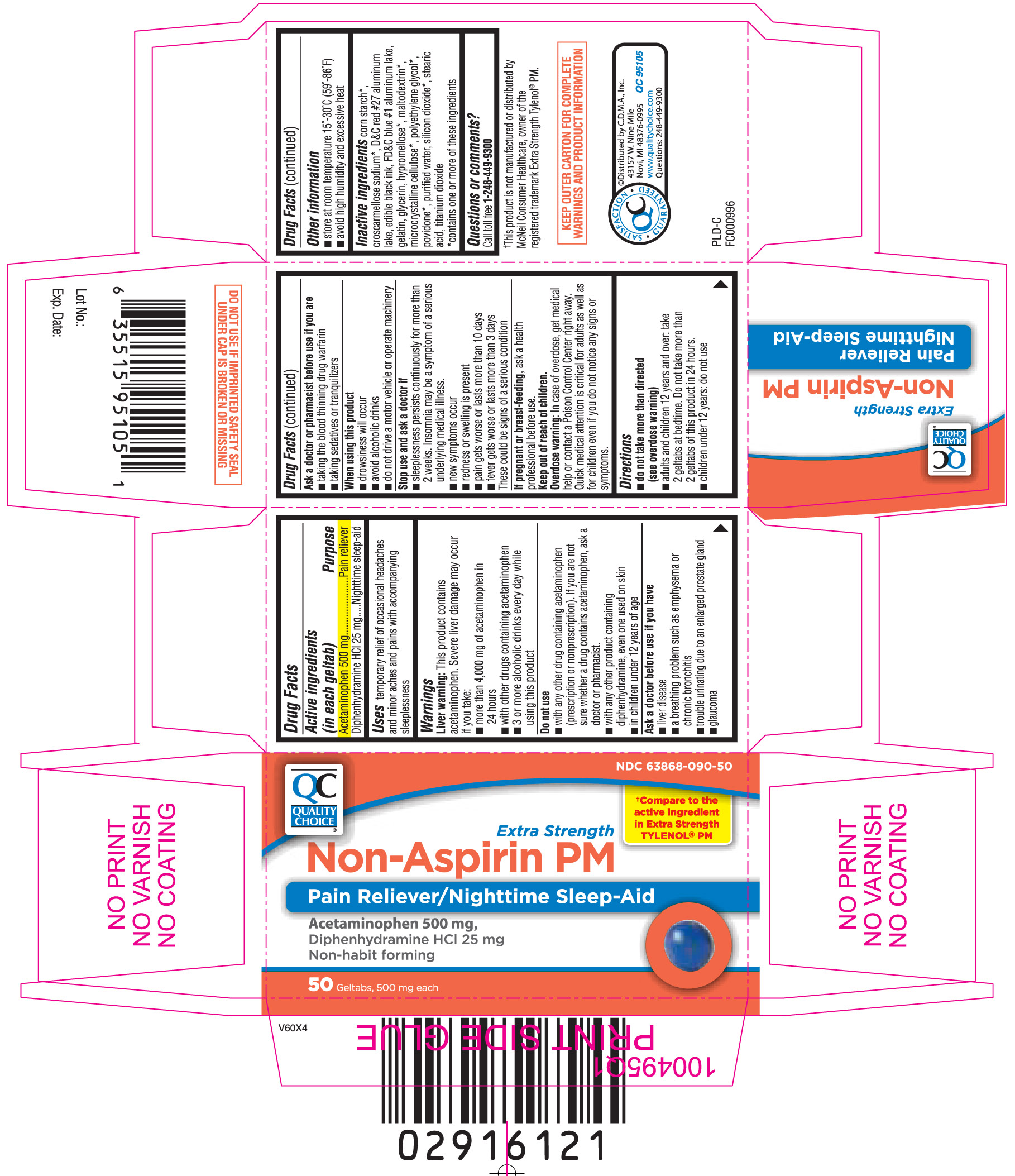
NON ASPIRIN PM EXTRA STRENGTH
-
acetaminophen and
diphenhydramine hydrochloride tablet, coated
QUALITY CHOICE (Chain Drug Marketing Association)
Acetaminophen 500 mg
Diphenhydramine HCl 25 mg
Pain reliever
Nighttime sleep-aid
temporary relief of occasional headaches and minor aches and pains with accompanying sleeplessness
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
These could be signs of a serious condition.
ask a health professional before use.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
corn starch*, croscarmellose sodium*, D&C red #27 aluminum lake, edible black ink, FD&C blue #1 aluminum lake, gelatin, glycerin, hypromellose*, maltodextrin*, microcrystalline cellulose*, polyethylene glycol*, povidone*, purified water, silicon dioxide*, stearic acid, titanium dioxide
*contains one or more of these ingredients
Call toll free 1-248-449-9300
†Compare to active ingredient in Extra Strength TYLENOL® PM
Extra Strength Non-Aspirin PM
Pain Reliever/Nighttime Sleep-Aid
Acetaminophen 500 mg,
Diphenhydramine HCl 25 mg
Non-habit forming
Geltabs, 500 mg each
†This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Extra Strength Tylenol® PM.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
©Distributed by C.D.M.A., Inc.
43157 W. Nine Mile
Novi, MI 48376-0995
Questions: 248-449-9300
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Quality Choice Acetaminophen PM Geltab
|
NON ASPIRIN PM
EXTRA STRENGTH acetaminophen, diphenhydramine hcl tablet, coated | ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH FINAL | part338 | 01/05/2023 | |
| Labeler - QUALITY CHOICE (Chain Drug Marketing Association) (011920774) |
| Registrant - P and L Development of New York Corporation (800014821) |