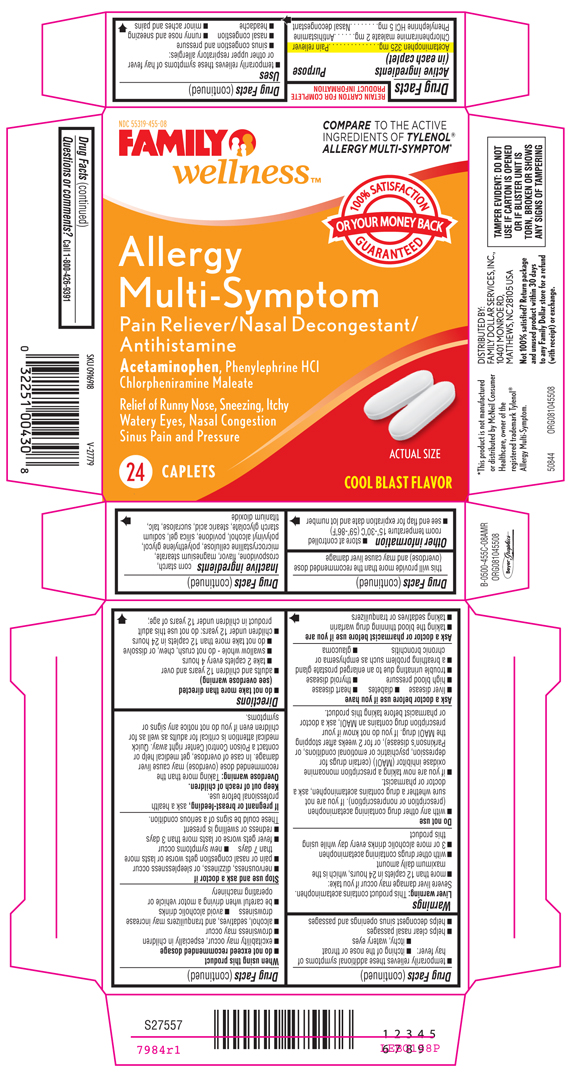
NON-ASPIRIN SINUS CONGESTION AND PAIN
-
acetaminophen,
chlorpheniramine maleate and
phenylephrine hydrochloride tablet
FRED'S, INC.
Acetaminophen 325 mg
Pain reliever
Antihistamine
Nasal decongestant
temporarily relieves these symptoms of hay fever or other upper respiratory allergies:
sinus congestion and pressure
nasal congestion
runny nose and sneezing
headache
minor aches and pains
temporarily relieves these additional symptoms of hay fever:
itching of the nose or throat
itchy, watery eyes
helps clear nasal passages
helps decongest sinus openings and passages
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
more than 12 caplets in 24 hours, which is the maximum daily amount
with other drugs containing acetaminophen
3 or more alcoholic drinks every day while using this product
with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
liver disease
diabetes
heart disease
high blood pressure
thyroid disease
trouble urinating due to an enlarged prostate gland
a breathing problem such as emphysema or chronic bronchitis
glaucoma
taking the blood thinning drug warfarin
taking sedatives or tranquilizers
do not exceed recommended dosage
excitability may occur, especially in children
drowsiness may occur
alcohol, sedatives, and tranquilizers may increase drowsiness
avoid alcoholic drinks
be careful when driving a motor vehicle or operating machinery
nervousness, dizziness, or sleeplessness occur
pain or nasal congestion gets worse or lasts more than 7 days
new symptoms occur
fever gets worse or lasts more than 3 days
redness or swelling is present
These could be signs of a serious condition.
ask a health professional before use.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
do not take more than directed (see overdose warning)
adults and children 12 years and over
take 2 caplets every 4 hours
swallow whole - do not crush, chew, or dissolve
do not take more than 12 caplets in 24 hours
children under 12 years: do not use this adult product in children under 12 years of age; this will provide more than the recommended dose (overdose) and may cause liver damage
store at controlled room temperature 15°-30°C (59°-86°F)
see end flap for expiration date and lot number
corn starch, crospovidone, flavor, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silica gel, sodium starch glycolate, stearic acid, sucralose, talc, titanium dioxide
Call 1-800-426-9391
FRED'S®
SEE NEW WARNINGS INFORMATION
Non-Aspirin
Sinus
Congestion & Pain
Pain Reliever/Nasal Decongestant/Antihistamine
Headache - Acetaminophen
Sinus Pressure
Nasal Congestion - Phenylephrine HCl 10 mg
Runny Nose - Chlorpheniramine maleate
Pseudoephedrine Free
24 Cool Caplets
with Cool Blast Flavor
Nighttime
Tested Against The Active Ingredient in: Congestion & Pain Nighttime TYLENOL® SINUS*
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Tylenol® Sinus Congestion & Pain.
50844 REV0810D45508
Fred's 44-455C
|
NON-ASPIRIN SINUS CONGESTION AND PAIN
acetaminophen, chlorpheniramine maleate and phenylephrine hcl tablet | ||||||||||||||||||||||
| ||||||||||||||||||||||
| ||||||||||||||||||||||
| ||||||||||||||||||||||
| ||||||||||||||||||||||
| ||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH NOT FINAL | part343 | 05/30/2006 | |
| Labeler - FRED'S, INC. (005866116) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 038154464 | PACK, PACK | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867894 | MANUFACTURE, MANUFACTURE | |