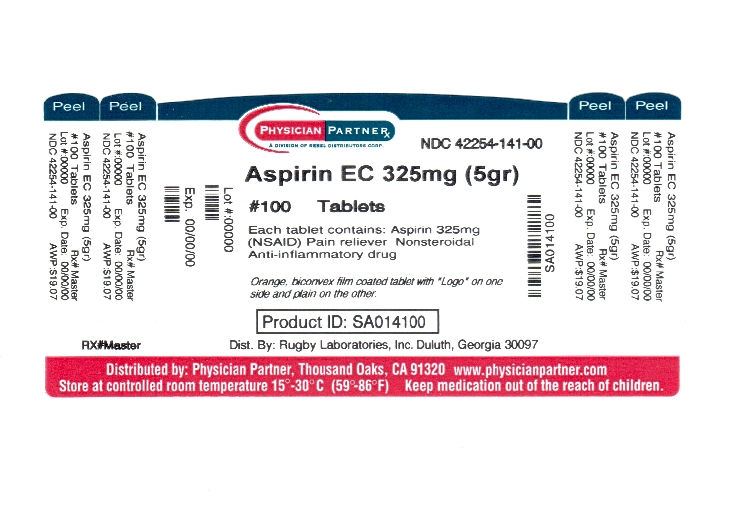
ENTERIC COATED ASPIRIN
-
aspirin tablet, delayed release
Rebel Distributors Corp
Active ingredient (in each tablet)
Aspirin 325 mg (NSAID*)
*nonsteroidal anti-inflammatory drug
Purpose
Pain reliever
Uses
temporarily relieves
Warnings
Reye's syndrome:
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert:
Aspirin may cause a severe allergic reaction which may include
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if the user
Do not use
if you have ever had an allergic reaction to any other pain reliever/fever reducer
Ask a doctor before use if
Ask a doctor or pharmacist before use if user is
Stop use and ask a doctor if
If pregnant or breast-feeding,
ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
DRINK A FULL GLASS OF WATER WITH EACH DOSE
Other information
Inactive ingredients
corn starch, croscarmellose sodium, D-C yellow #10 aluminum lake, FD-C yellow #6 aluminum lake, hypromellose, methacrylic acid copolymer, microcrystalline cellulose, mineral oil, polysorbate 80, simethicone, sodium hydroxide, sodium lauryl sulfate, talc, titanium dioxide, triethyl citrate
Questions or comments?
call
1-800-645-2158, 9 am - 5 pm ET, Monday - Friday
|
ENTERIC COATED ASPIRIN
aspirin tablet, delayed release | ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part343 | 09/20/2011 | |
| Labeler - Rebel Distributors Corp (118802834) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Rebel Distributors Corp | 118802834 | RELABEL, REPACK | |