ASPIRIN
-
aspirin tablet, coated
TIME CAP LABS INC
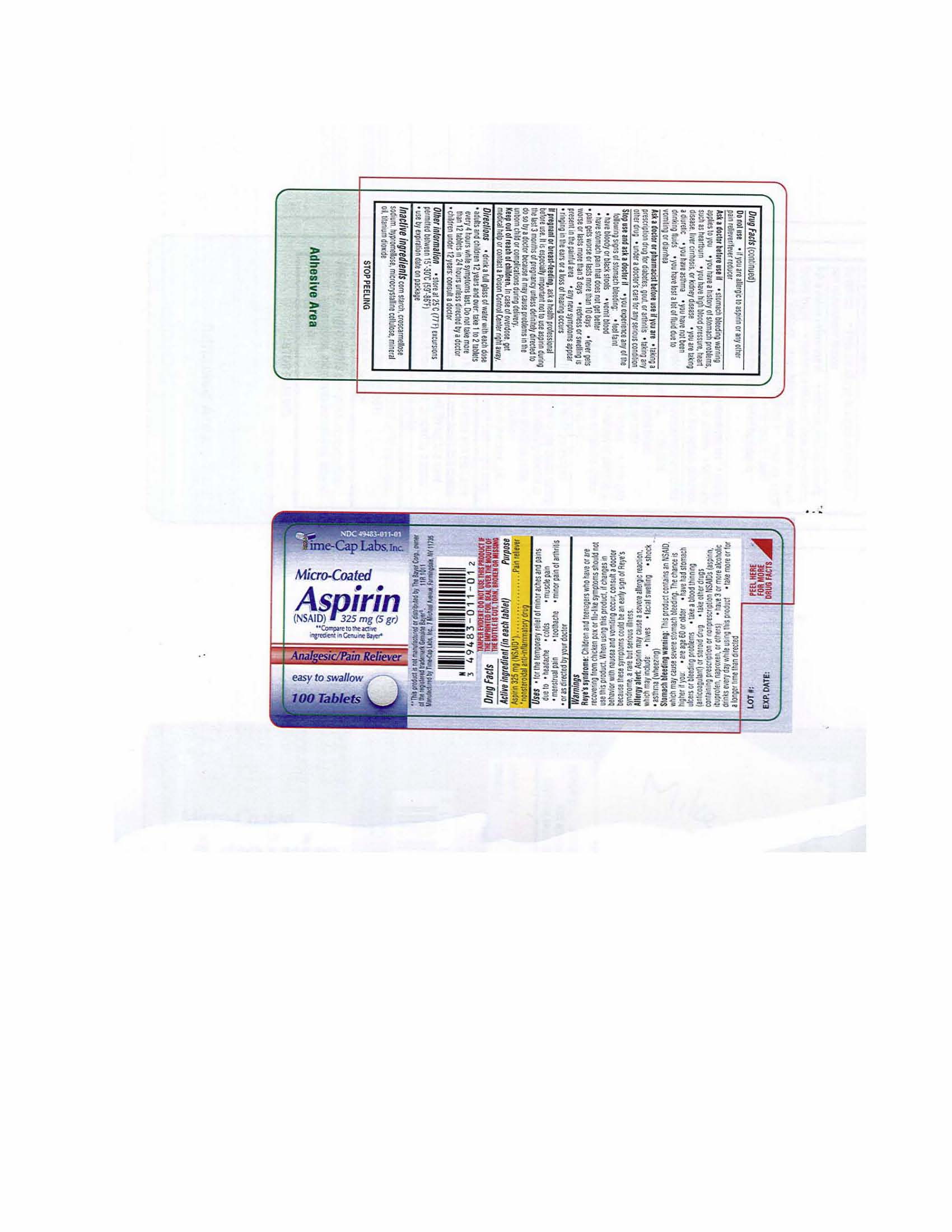
Each tablet contains Aspirin 325 mg
corn starch, croscarmellose sodium, Hypromellose, microcrystalline cellulose, mineral oil, titanium dioxide
WARNINGS: Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include, hives, facial swelling, shock, asthma (wheezing)
Stomach bleeding warning: This product contains an NSAID which may cause severe stomach bleeding. The chance is higher if you are age 60 or older, have had stomach ulcers or bleeding problems, take a blood thinning (anticoagulant) or steroid drug, take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others); have 3 or more alcoholic drinks every day while using this product, take more or for a longer time than directed
Do not use
If you are allergic to aspirin or any other pain reliever/fever reducer
Directions:
Drink a full glass of water with each dose
Adults and children 12 years and over: take 1 to 2 tablets every 4 hours while symptoms last
Do not take more than 12 tablets in 24 hours unless directed by a doctor.
Children under 12 years: consult a doctorKeep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Uses:
For the temporary relief of minor aches and pains due to:
headache, colds, muscle pain, menstrual pain, toothache, minor pain of arthritis or as directed by your doctor.
|
ASPIRIN
aspirin tablet, coated | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part343 | 04/18/2012 | |
| Labeler - TIME CAP LABS INC (037052099) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| TIME CAP LABS INC | 037052099 | manufacture | |