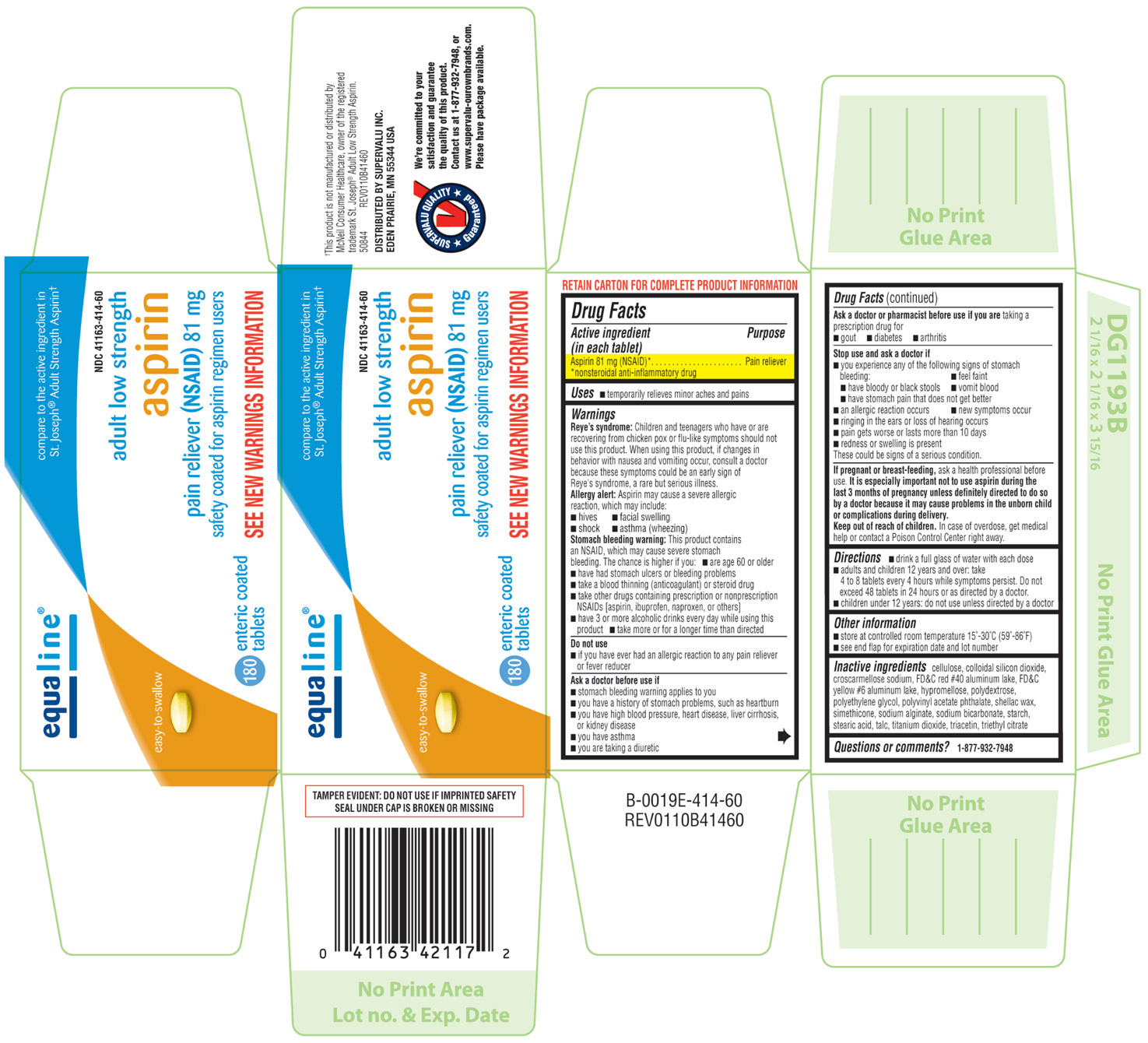
ADULT LOW STRENGTH ASPIRIN
-
aspirin tablet, coated
SUPERVALU INC.
Aspirin 81 mg (NSAID)*
*nonsteroidal anti-inflammatory drug
Pain reliever
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include:
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
taking a prescription drug for
These could be signs of a serious condition.
ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause prblems in the unborn child or complications during delivery.
In case of overdose, get medical help or contact a Poison Control Center right away.
cellulose, colloidal silicon dioxidem croscarmellose sodium, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, shellac wax, simethicone, sodium alginate, sodium bicarbonate, starch, stearic acid, talc, titanium dioxide, triacetin, triethyl citrate
1-877-932-7948
The product packaging shown below represents a sample of that currently in use. Additional packaging may also be available.
equaline®
Compare to the active ingredient in
St. Joseph® Adult Strength Aspirin
NDC 41163-414-60
adult low strength
aspirin
pain reliever (NSAID) 81 mg
safety coated for aspirin regimen users
easy-to-swallow
180 enteric coated tablets
SEE NEW WARNINGS INFORMATION
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
50844 REV 0110B414460
DISTRIBUTED BY SUPERVALU INC.
EDEN PRAIRIE, MN 55344 USA
Product Label
|
ADULT LOW STRENGTH ASPIRIN
aspirin tablet, coated | ||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH NOT FINAL | part343 | 01/14/2004 | |
| Labeler - SUPERVALU INC. (006961411) |