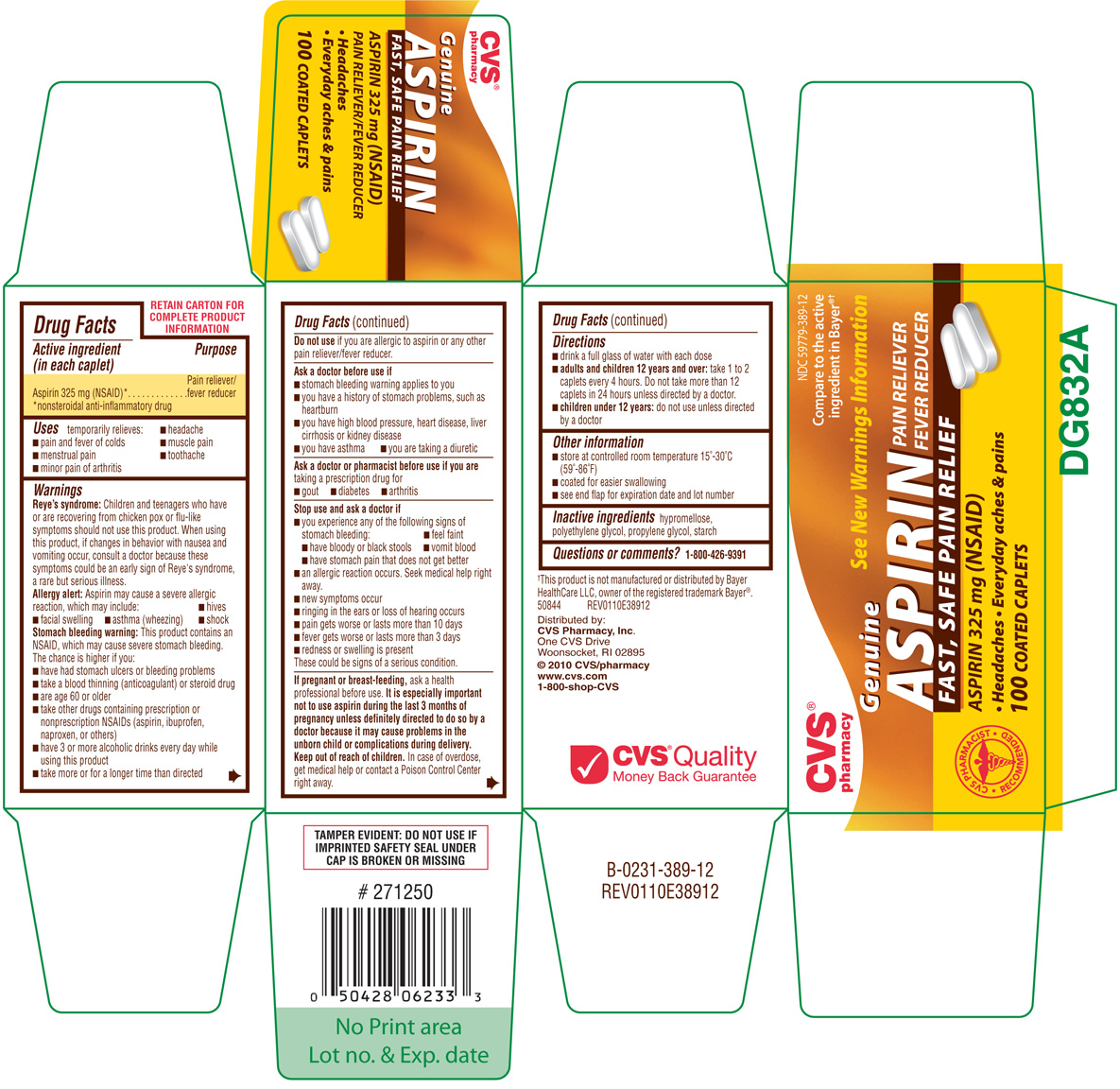
GENUINE ASPIRIN
-
aspirin tablet
WOONSOCKET PRESCRIPTION CENTER, INCORPORATED
(in each caplet)
Aspirin 325 mg (NSAID)*
Pain reliever/fever reducer
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because theses symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Aspirin may cause a severe allergic reaction, which may include:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
if you are allergic to aspirin or any other pain reliever/fever reducer.
taking a prescription drug for
These could be signs of a serious condition.
ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
In case of overdose, get medical help or contact a Poison Control Center right away.
hypromellose, polyethylene glycol, propylene glycol, starch
1-800-426-9391
The product packaging shown below represents a sample of that currently in use. Additional packaging may also be available.
CVS®
pharmacy
NDC 59779-389-12
Compare to the active
ingredient in Bayer*†
See new Warnings Information
Genuine
Aspirin
PAIN RELIEVER
FEVER REDUCER
FAST, SAFE PAIN RELIEF
ASPIRIN 325 mg (NSAID)
● Headaches ● Everyday aches & pains
100 COATED CAPLETS
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
50844 REV0110E38912
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
© 2010 CVS/pharmacy
1-800-shop-CVS
|
GENUINE ASPIRIN
aspirin tablet | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part343 | 05/17/2001 | |
| Labeler - WOONSOCKET PRESCRIPTION CENTER, INCORPORATED (062312574) |
| Registrant - L.N.K. International, Inc. (832867837) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| L.N.K. International, Inc | 832867894 | MANUFACTURE | |