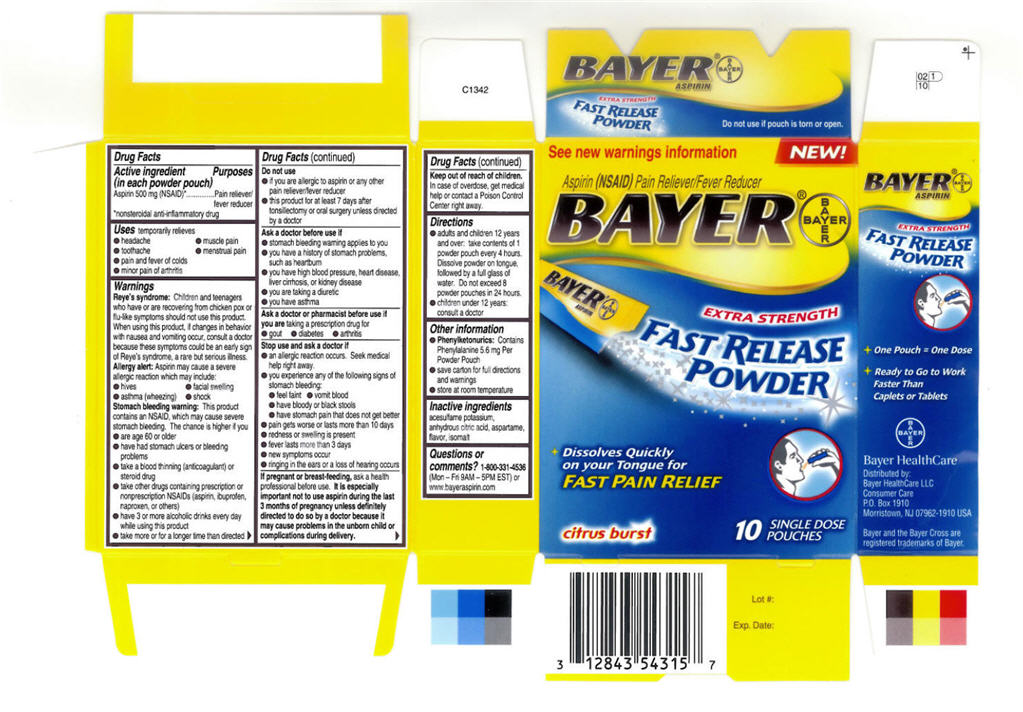
BAYER FAST RELEASE
-
aspirin powder
Bayer Corporation Consumer Care Division
Drug Facts
Aspirin 500 mg (NSAID)1
Pain reliever/fever reducer
temporarily relieves
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product.
When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Aspirin may cause a severe allergic reaction which may include:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
Do not use
Ask a doctor before use if
Ask a doctor or pharmacist before use if you are taking a prescription drug for
Stop use and ask a doctor if
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
acesulfame potassium, anhydrous citric acid, aspartame, flavor, isomalt
1-800-331-4536 (Mon – Fri 9AM – 5PM EST) or www.bayeraspirin.com
See new warings information
NEW!
Aspirin (NSAID) Pain Reliever/Fever Reducer
BAYER®
EXTRA STRENGTH
FAST RELEASE
POWDER
Dissolves Quickly
on your Tongue for
FAST PAIN RELIEF
citrus burst
10 SINGLE DOSE
POUCHES
|
BAYER
FAST RELEASE aspirin powder | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH FINAL | part341 | 06/30/2010 | |
| Labeler - Bayer Corporation Consumer Care Division (785159372) |